
Implementing the EU Chemicals Strategy for Sustainability: The case of food contact chemicals of concern - ScienceDirect

Chapter 5 Welcome to the Schengen Hotel in: Immigration and Privacy in the Law of the European Union
Commission Delegated Regulation (EU) 2016/161 of 2 October 2015 supplementing Directive 2001/83/EC of the European Parliament an

Pest categorisation of the Ralstonia solanacearum species complex - - 2019 - EFSA Journal - Wiley Online Library
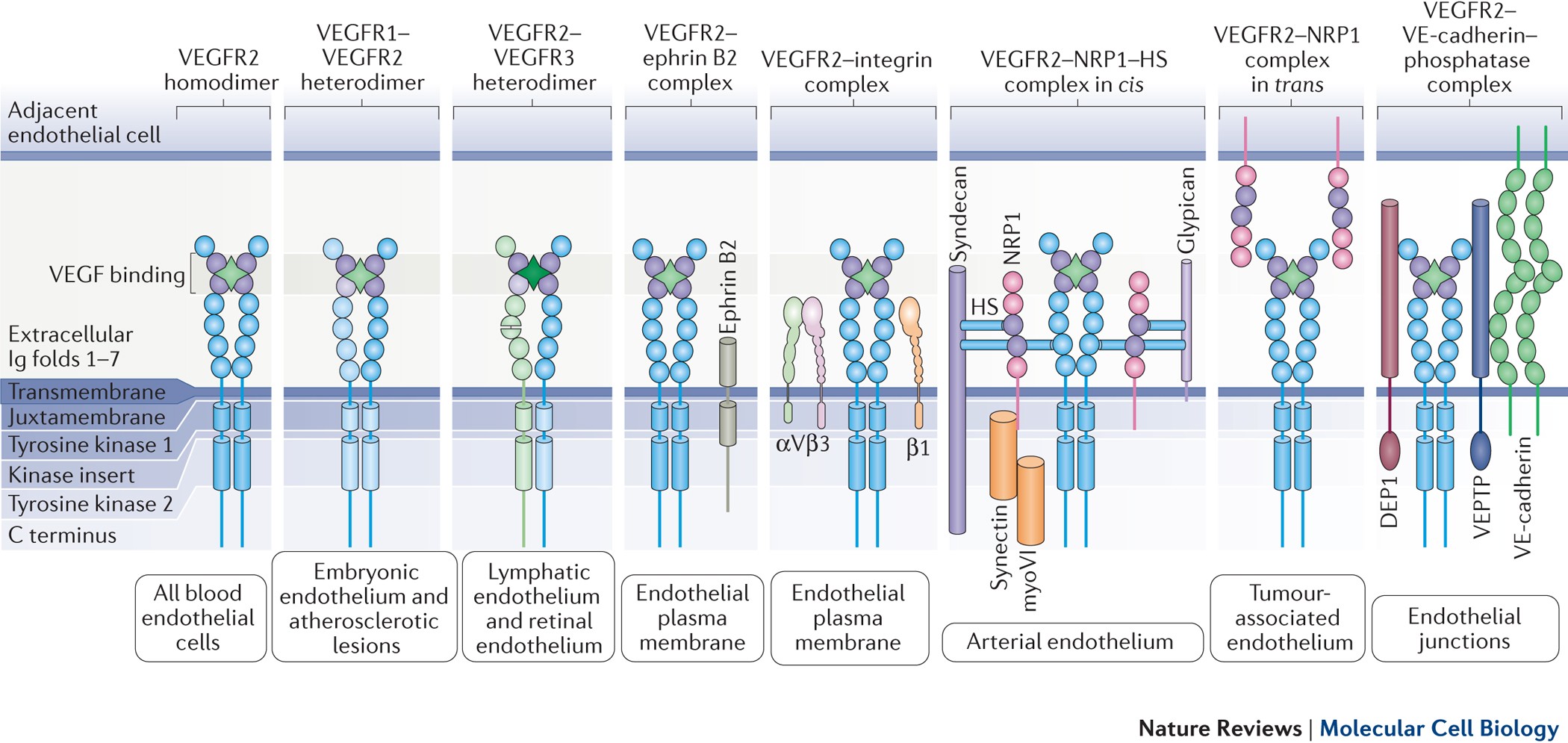
Mechanisms and regulation of endothelial VEGF receptor signalling | Nature Reviews Molecular Cell Biology
ESSD - The consolidated European synthesis of CH4 and N2O emissions for the European Union and United Kingdom: 1990–2019
11107/16 MBT/sy DG C 1 Delegations will find attached document C(2016) 4167 final (Kenya). Encl.: C(2016) 4167 final
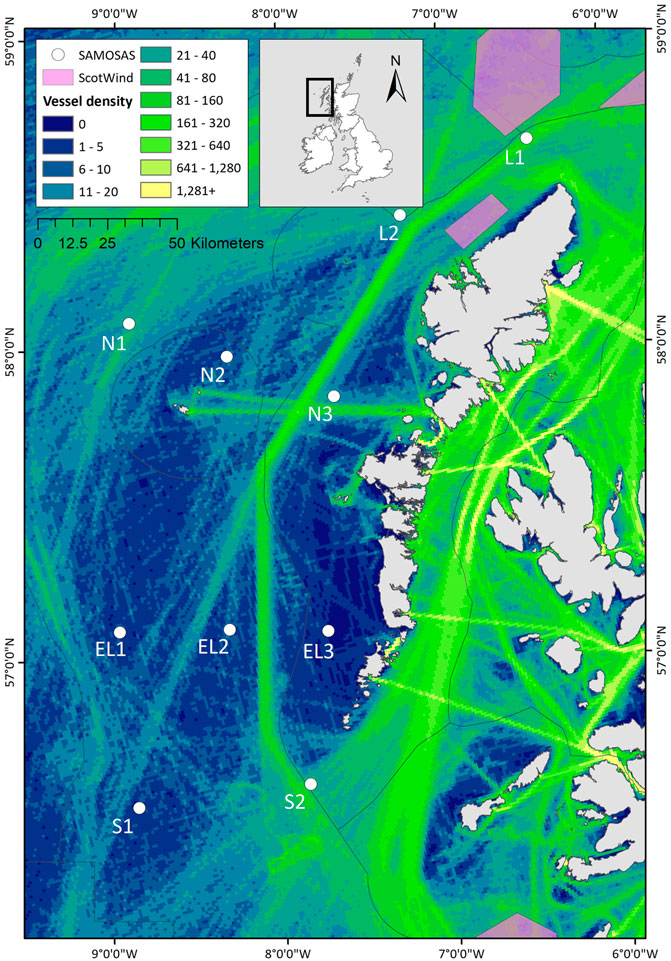
Frontiers | Monitoring cetacean occurrence and variability in ambient sound in Scottish offshore waters
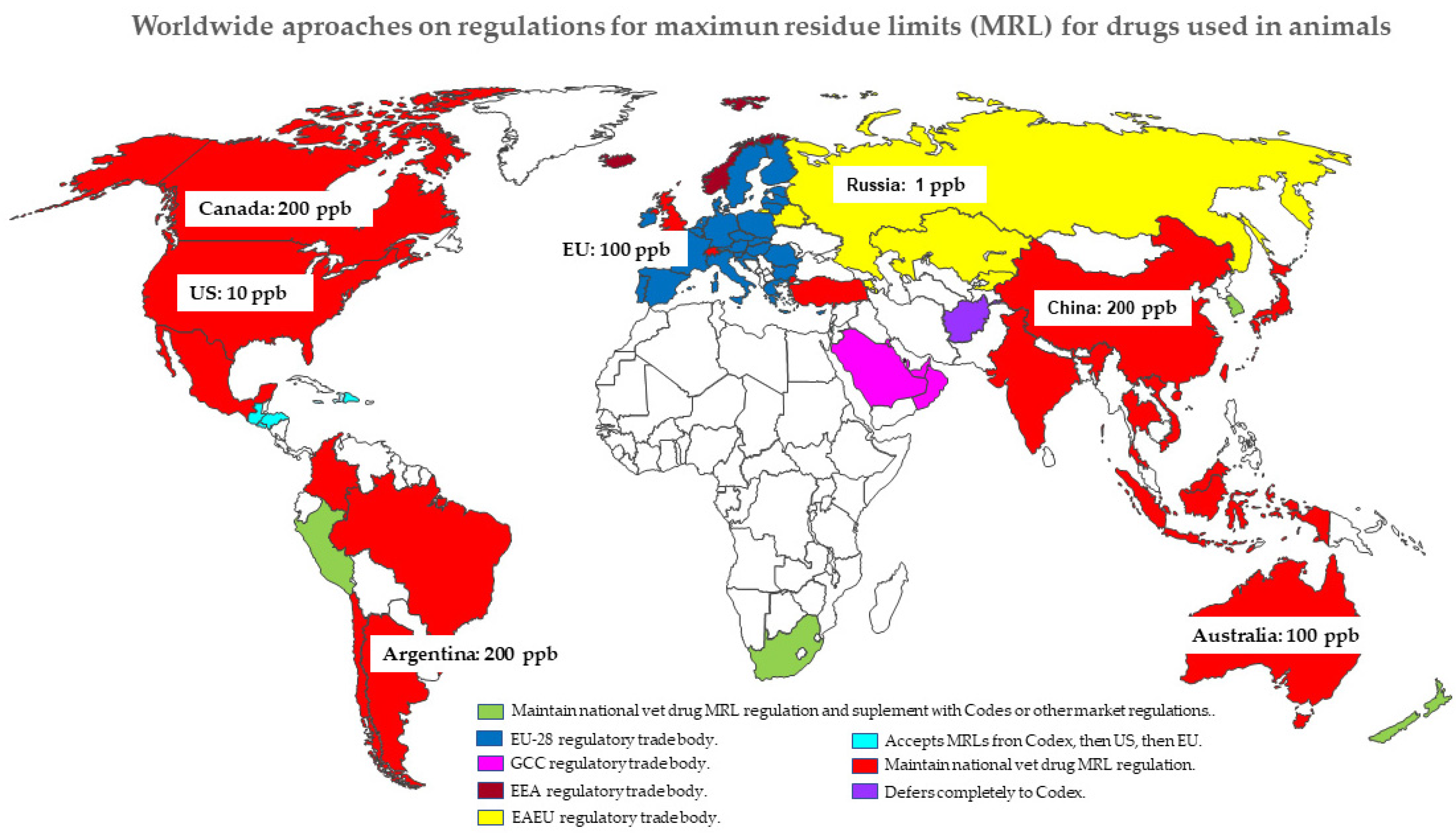
Animals | Free Full-Text | Rational Pharmacotherapy in Infectious Diseases: Issues Related to Drug Residues in Edible Animal Tissues
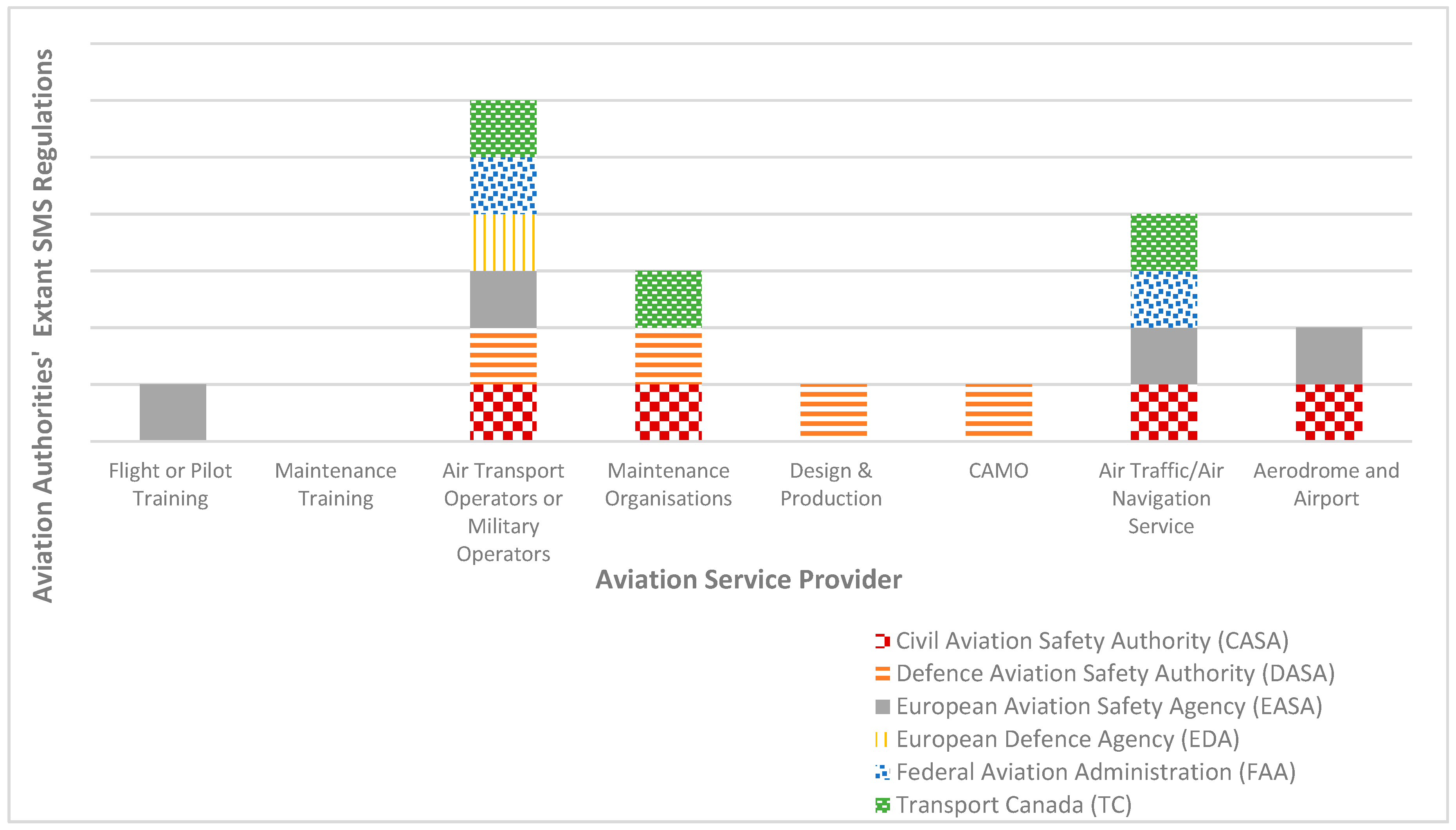
Aerospace | Free Full-Text | The Regulatory Framework for Safety Management Systems in Airworthiness Organisations
Commission notice – Application of the Union's pharmaceutical acquis in markets historically dependent on medicines supply f
COUNCIL REGULATION (EU) 2019/ 999 - of 13 June 2019 - amending Regulation ( EU) No 1387 / 2013 suspending the a









